When there is a mass shooting, we not only treat the wounded and bury the dead, but investigate about the shooter/shooters, what kind of weapons are used, how he/she/ they obtain the weapons, who else provided material help and all related questions. So why the media do not ask such questions about the formation of hurricane and other storms hit nations around the world is beyond me.
The last time there was this much carbon dioxide (CO2) in the Earth’s atmosphere, modern humans didn’t exist. Megatoothed sharks prowled the oceans, the world’s seas were up to 100 feet higher than they are today, and the global average surface temperature was up to 11°F warmer than it is now.
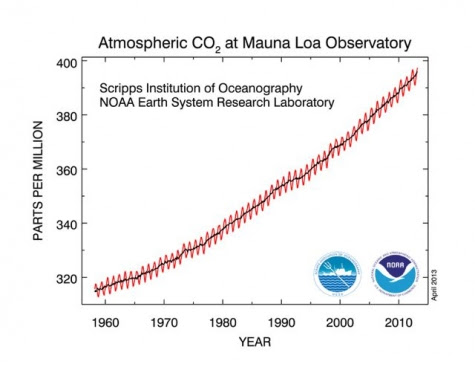
As we near the record for the highest CO2 concentration in human history — 400 parts per million — climate scientists worry about where we were then, and where we’re rapidly headed now.
According to the data gathered at the Mauna Loa Observatory in Hawaii, the 400 ppm mark may briefly be exceeded this month, when CO2 typically hits a seasonal peak in the Northern Hemisphere, although it is more likely to take a couple more years until it stays above that threshold, according to Ralph Keeling, a researcher at the Scripps Institute of Oceanography.
Click image to enlarge. Credit: Scripps Institution of Oceanography.
CO2 levels are far higher now than they have been for anytime during the past 800,000 years.
Keeling is the son of Charles David Keeling, who began the CO2 observations at Mauna Loa in 1958 and for whom the iconic “Keeling Curve” is named.
Carbon dioxide is the most important long-lived global warming gas, and once it is emitted by burning fossil fuels such as coal and oil, a single CO2 molecule can remain in the atmosphere for hundreds of years. Global CO2 emissions reached a record high of 35.6 billion tonnes in 2012, up 2.6 percent from 2011.
Carbon dioxide and other greenhouse gases warm the planet by absorbing the sun’s energy and preventing heat from escaping back into space.
The news that CO2 is near 400 ppm for the first time highlights a question scientists have been investigating using a variety of methods: when was the last time that CO2 levels were this high, and what was the climate like back then?
There is no single, agreed-upon answer to those questions as studies show a wide date range from between 800,000 to 15 million years ago. The most direct evidence comes from tiny bubbles of ancient air trapped in the vast ice sheets of Antarctica. By drilling for ice cores and analyzing the air bubbles, scientists have found that, at no point during at least the past 800,000 years have atmospheric CO2 levels been as high as they are now.
That means that in the entire history of human civilization, CO2 levels have never been this high.
Credit: NOAA.
Other research, though, shows that you have to go back much farther in time, well beyond 800,000 years ago, to find an instance where CO2 was sustained at 400 ppm or greater.
For a 2009 study, published in the journal Science, scientists analyzed shells in deep sea sediments to estimate past CO2 levels, and found that CO2 levels have not been as high as they are now for at least the past 10 to 15 million years, during the Miocene epoch.
“This was a time when global temperatures were substantially warmer than today, and there was very little ice around anywhere on the planet. And so sea level was considerably higher — around 100 feet higher — than it is today,” said Pennsylvania State University climate scientist Michael Mann, in an email conversation. “It is for this reason that some climate scientists, like James Hansen, have argued that even current-day CO2 levels are too high. There is the possibility that we’ve already breached the threshold of truly dangerous human influence on our climate and planet.”
Sea levels are increasing today in response to the warming climate, as ice sheets melt and seas expand due to rising temperatures. Scientists are projecting up to 3 feet or more of global sea level rise by 2100, which would put some coastal cities in peril.
While there have been past periods in Earth’s history when temperatures were warmer than they are now, the rate of change that is currently taking place is faster than most of the climate shifts that have occurred in the past, and therefore it will likely be more difficult to adapt to.
A 2011 study in the journal Paleoceanography found that atmospheric CO2 levels may have been comparable to today’s as recently as sometime between 2 and 4.6 million years ago, during the Pliocene epoch, which saw the arrival of Homo habilis, a possible ancestor of modern homo sapiens, and when herds of giant, elephant-like Mastadons roamed North America. Modern human civilization didn’t arrive on the scene until the Holocene Epoch, which began 12,000 years ago.
Regardless of which estimate is correct, it is clear that CO2 levels are now higher than they have ever been in mankind’s history. With global CO2 emissions continuing on an upward trajectory that is likely to put CO2 concentrations above 450 ppm or higher, it is extremely unlikely that the steadily rising shape of the Keeling Curve is going to change anytime soon.
“There’s an esthetic to the curve that’s beautiful science and troubling reality,” Keeling said. “I’d very much like to see the curve change from going steadily upward to flattening out.”
Related Content
CO2 Emissions Expected to Rise Significantly By 2030
Global Carbon Emissions Hit Record High
Global CO2 Levels Set to Pass 400 ppm Milestone
In the Curve: Monitoring Rising Carbon Emissions
A tree can absorb as much as 48 pounds of carbon dioxide per year and can sequester 1 ton of carbon dioxide by the time it reaches 40 years old.Through burning fossil fuels, humans are rapidly driving up levels of carbon dioxide in the atmosphere, which in turn is raising global temperatures.
But not all the CO2 released from burning coal, oil and gas stays in the air. Currently, about 25% of the carbon emissions produced by human activity are absorbed by plants, and another similar amount ends up in the ocean.
To know how much more fossils fuels we can burn while avoiding dangerous levels of climate change, we need to know how these “carbon sinks” might change in the future. A new study led by Dr. Sun and colleagues published in the US journal Proceedings of the National Academy of Sciences shows the land could take up slightly more carbon than we thought.
But it doesn’t change in any significant way how quickly we must decrease carbon emissions to avoid dangerous climate change.
Models overestimate CO2
The new study estimates that over the past 110 years some climate models over-predicted the amount of CO2 that remains in the atmosphere, by about 16%.
Models are not designed to tell us what the atmosphere is doing: that’s what observations are for, and they tell us that CO2 concentrations in the atmosphere are currently over 396 parts per million, or about 118 parts per million over pre-industrial times.
These atmospheric observations are in fact the most accurate measurements of the carbon cycle.
But models, which are used to understand the causes of change and explore the future, often don’t match perfectly the observations. In this new study, the authors may have come up with a reason that explains why some models overestimate CO2 in the atmosphere. https://www.thoughtco.com/whic
“Human influence on the climate system is clear, and recent anthropogenic emissions of green-house gases are the highest in history. Recent climate changes have had widespread impacts on human and natural systems. “Warming of the climate system is unequivocal, and since the 1950s, many of the observed changes are unprecedented over decades to millennia. The atmosphere and ocean have warmed, the amounts of snow and ice have diminished, and sea level has risen.” (2014)
International academies joint statement: Global response to climate change
“Climate change is real. There will always be uncertainty in understanding a system as complex as the world’s climate. However, there is now strong evidence that significant global warming is occurring.” (2005, national academies of science of Brazil, China, France, Germany, India, Italy, Japan, Russia, United Kingdom, United States)
U.S. Global Change Research Program: Highlights of the Findings of the U.S. Global Change Research Program Climate Science Special Report
“Based on extensive evidence, … it is extremely likely that human activities, especially emissions of greenhouse gases, are the dominant cause of the observed warming since the mid-20th century. For the warming over the last century, there is no convincing alternative explanation supported by the extent of the observational evidence.
“In addition to warming, many other aspects of global climate are changing, primarily in response to human activities. Thousands of studies conducted by researchers around the world have documented changes in surface, atmospheric, and oceanic temperatures; melting glaciers; diminishing snow cover; shrinking sea ice; rising sea levels; ocean acidification; and increasing atmospheric water vapor.” (November 2017)
U.S. National Academy of Sciences: Understanding and Responding to Climate Change
“The scientific understanding of climate change is now sufficiently clear to begin taking steps to prepare for climate change and to slow it.” (2008)
Why are Rainforests Disappearing
The tropical and subtropical regions are most vulnerable areas, that maintain a large part of biodiversity on the planet. There, moist forest have been razed by loggers and bulldozers. The use of these lands for agricultural crops and the breeding of heat-resistant livestock, as well as the use of natural resources for various purposes (timber, textile, pharmaceutical and other industries) resulted in the progressive devastation of such areas.
The worlds rain forests are vital to sustaining life and keeping the ecological balance so crucial to survival for all beings. As they become more and more depleted, the futures uncertainty becomes a dark vision that cannot be ignored. Mans own ignorance and greed has greatly jeopardized the future of life on earth as we know it and without drastic changes in the here and now, the future is nothing short of dismal.
The metabolism of cancer is approximately eight times greater than the metabolism of normal cells (that’s why they love sugar so much), however, Warburg forgot to tell the world—not only are the oxygen levels low but so are carbon dioxide (CO2) levels. And he did not tell a soul that by breathing too fast (as most people do) they are getting rid of too much CO2 and that is what is driving down the oxygen levels to the point that cells turn cancerous.
Otto Warburg was telling us that the cellular metabolism of cancer cells matches closely those of yeast or mold or fungus—that is, the cells ferment sugar/glucose/dextrose rather than oxidize it via the cellular mitochondria. So it follows logically that the same medical approach that successfully targets cancer would do the same for these yeasts, molds and fungus.
Warburg had only part of the story and no one has put the finishing chapter in so we can finally come to rest with a full understanding and appreciation of what we are facing when cancer knocks on our door. Natural Allopathic Medicine approaches the core problem of oxygen from many directions at once.
Dr. Lesley Walker, Cancer Research UK’s director of cancer information, said, “For a long time scientists have been looking for ways to boost the oxygen supply to tumors to improve response to treatment.” He was referring to treatment with radiation, but this would apply very much to a whole range of natural treatments.
If lack of oxygen is a key driver of cancer growth then so is low CO2, pH and depressed cell voltage.
Some doctors have used the observations of Otto Warburg to treat certain forms of cancer as the human body’s reaction to a fungal infection. (See Dr. Tullio Simoncini “Cancer is a Fungus”). By using simplistic anti-fungal substances such as sodium bicarbonate (baking soda) for internal cancers, and tincture of iodine for external cancers, Dr. Simoncini has successfully treated many patients without adverse side effects. http://drsircus.com/cancer/the
Several studies from England and the USA found that breathing various carbogen mixtures significantly improves oxygenation of tumors. The general opinion of these researchers is that
“Perfusion insufficiency and the resultant hypoxia are recognized as important mechanisms of resistance to anticancer therapy. …”(Powell et al, 1997).
A large group of British scientists from the Paul Strickland Scanner Centre revealed that when 14 cancer patients breathed various carbogen mixtures (with 2%, 3.5% and 5% CO2 content, the rest was O2) “arterial oxygen tension increased at least three-fold from basal values” (Baddeley et al, 2000). They also found that
“…The results suggest that 2% CO2 in O2 enhances arterial O2 levels to a similar extent as 3.5% and 5% CO2 and that it is well tolerated” (Baddeley et al, 2000).
Another group of British researchers directly measured oxygen pressure in cancer cells and concluded,
“This study confirms that breathing 2% CO2 and 98% oxygen is well tolerated and effective in increasing tumor oxygenation” (Powell et al, 1999).These results generate the following question. Which gas, CO2 or O2 is the main contributor to increased oxygenation of cells and by how much? The amounts of both gases in mixtures were much higher than the amounts of O2 and CO2 in normal air.
Let us, first, consider the influence of O2. There are two O2 states in the arterial blood: O2 that is combined with hemoglobin or red blood cells and O2 that is dissolved in blood plasma. Which component produces the main increase in oxygenation of the arterial blood?
As we considered in Chapter 1, the saturation of hemoglobin wit h O2 under normal conditions (or when breathing normal air) is about 98%. Increased O2 pressure can raise this value to almost 100%. This would cause about a 2% increase in the arterial blood in comparison with the initial value. In addition, when patients breathe carbogen mixtures more O2 can be freely dissolved in blood plasma (this O2 is not bound to red blood cells).
In normal conditions the contribution of dissolved O2 is about 1.5% of the total blood O2 as the remaining 98.5% O2 is combined with hemoglobin. Increasing O2 content in the inspired air (almost five times) can increase freely dissolved oxygen to about 6-7% in relation to the initial value (1.5%).
Hence, increasing the O2 component in the inspired air (from about normal 20% up to almost 100%) can cause about 8-9% increase in total O2 content in the arterial blood (with 2% increase for combined oxygen and 6-7% for freely dissolved oxygen).
British professionals decided “to assess the relative contributions of carbon dioxide and oxygen to this response and the tumor oxygenation state, the response of GH3 prolactinomas to 5% CO2/95% air, carbogen and 100% O2” (Baddeley et al, 2000).
That was done using magnetic resonance imaging and PO2 histography. They found that,
“A 10-30% image intensity increase was observed during 5% CO2/95% air breathing, consistent with an increase in tumor blood flow, as a result of CO2-induced vasodilation… A small 5-10% increase was observed in response to 100% oxygen, highlighting the dominance of CO2-induced vasodilation in the carbogen response” (Baddeley et al, 2000).
It is not oxygen, but carbon dioxide that is also the main substance responsible for the main improvement in oxygenation of tumors, while high O2 concentrations, while providing additional improvement in oxygenation of the arterial blood and tumors, are toxic for lungs alveoli due to high reactivity of oxygen and resulting oxidative damage to tissues and formation of free radicals.
Hence, it is logical to expect that if the same patients use 100% O2 for many days, not just hours, the oxidative damage can produce more harm for the whole body than the benefits of pure oxygen for tumor reduction. Higher CO2, on the other hand, will cause sustained improvements in tumor oxygenation without any negative effects. (In fact, there are many more positive effects due to various uses of CO2 in the human body.)
Indeed, more detailed analysis or dynamic of improved oxygenation was investigated by German scientists (the abstract is one paragraph below; Thews et al, 2002). They found that positive effects of pure oxygen are very short in duration, while CO2 produces lasting improvement.
But a cancer patient can safely increase own CO2 content in the body and oxygenation of the tumors naturally by learning how to breathe less (Section “Learn here“), while preserving normal arterial blood oxygenation (about 98% for O2 hemoglobin saturation) and avoiding oxidative damage, expensive therapies and equipment, hours and hours of exhaustive labor has been done by medical professionals.




