
Gut microbiome changes linked to onset of clinically evident rheumatoid arthritis
Story by Science X staff
Changes in the make-up of the gut microbiome are linked to the onset of clinically evident rheumatoid arthritis in those at risk of the disease because of genetic, environmental, or immunological factors, suggests research published online in the Annals of the Rheumatic Diseases.
It’s not clear if this instability is a cause or consequence of disease development, emphasize the researchers, but the findings might nevertheless help to identify those at risk as well as paving the way for preventive and personalized treatment strategies, they suggest.
Previously published research consistently shows an unfavorable imbalance in the gut microbiomes of those at risk as well as those diagnosed with rheumatoid arthritis compared with the gut microbiomes of those without the disease.
But it’s not clear exactly which microbes might be involved.
To explore this further, the researchers tracked changes in the gut microbiome profiles of 124 people at risk of developing rheumatoid arthritis; in seven newly diagnosed people; and in 22 healthy people over a period of 15 months, by assessing their stool and blood samples at five different time points.
Those at risk were identified by the presence of precursor anti cyclic citrullinated protein (anti-CCP) antibodies, which attack healthy cells and are specific for rheumatoid arthritis, and by experience of joint pain in the preceding three months.
Weekly dietary intake was similar among all three groups, although alcohol intake and the amount of regular moderate to vigorous exercise taken differed.
During the study period, 30 of the 124 in the at risk group progressed to rheumatoid arthritis, and their microbial diversity was notably reduced compared with that of the healthy comparison group, particularly within specific areas—known as alpha diversity.
Alpha diversity was also reduced in both those who progressed and those who didn’t, and linked to anti-CCP antibody levels. In those with low anti-CCP antibody levels, microbial diversity was comparable with that of the healthy comparison group.
Recognized genetic, blood, and imaging risk factors for arthritis development were also significantly linked to lower microbial diversity, as was steroid use.
A specific strain of Prevotellaceae sp—(ASV2058) most likely P. copri—was abundant in the microbiomes of those who progressed as well as in those of the newly diagnosed, but not in the microbiomes of those in the healthy comparison group.
Another strain (ASV1867) of P. copri was also increased at the start of the study in those who progressed, possibly suggesting that different strains of P. copri might have different roles in rheumatoid arthritis progression, say the researchers.
Further analysis indicated that both enrichment (three) and depletion (five) of Prevotellaceae-specific strains were associated with progression.
While P. copri strains were most strongly associated with clinical risk factors for rheumatoid arthritis, other Prevotellaceae strains were also implicated, including Alloprevotella, Paraprevotella clara, Prevotella stercorea, Prevotellamassilia timonensis and Prevotella shahii.
The greatest instability in gut microbiome profile was seen among those who developed arthritis up to 10 months before diagnosis. But this profile was relatively stable in those diagnosed after this time—10–15 months before the development of rheumatoid arthritis. This suggests that changes in the gut microbiome are a late stage phenomenon, say the researchers.
This is an observational study, precluding any firm conclusions to be drawn about causal factors. And the researchers acknowledge various study limitations, including the small number of participants, the relatively short monitoring period, and the lack of direct one on one comparison between the at-risk and healthy participants.
But they conclude, “Individuals at risk of [rheumatoid arthritis] harbor a distinctive gut microbial composition, including but not limited to an overabundance of Prevotellaceae species. This microbial signature is consistent and correlates with traditional risk factors.
“Longitudinal examination shows a dynamic microbial environment preceding [rheumatoid arthritis] onset. Further research into this late phase of disease development is merited, especially given the potential of the gut microbiome as a target for prevention, including in high-risk individuals with imminent arthritis.”
More information: Dynamics of the gut microbiome in individuals at risk of rheumatoid arthritis: a cross-sectional and longitudinal observational study, Annals of the Rheumatic Diseases (2024). DOI: 10.1136/ard-2024-226362
Provided by British Medical Journal
Dysbiosis means that you have an imbalance in the different types of microscopic organisms living in your body. If there are too many of some types and not enough of others, they don’t work with you as they should, and they might work against you. In particular, dysbiosis in your gut may have broad effects on your health.
What is dysbiosis?
Dysbiosis is an imbalance within a community of microorganisms living together — a microbiome. Our bodies are host to several distinct microbiomes — communities of microorganisms that live with us and assist us in various ways. A balanced microbiome is one where there’s a healthy diversity of microorganisms, where no single bacteria, virus or fungus dominates. Dysbiosis means there’s a lack of diversity and pH balance. When they’re imbalanced, it changes how they function in your body.
How does dysbiosis affect me?
In any microbiome, a lack of diversity and balance can pave the way for one type of microorganism to take over. Dysbiosis makes us more vulnerable to infections from germs living inside and outside of our bodies. It can also interfere with other important services that our microbiomes normally provide for us. For instance, your gut microbiome provides you with many services by interacting with your body in a multitude of ways.
What can gut dysbiosis lead to?
Bacterial dysbiosis in your gut is directly involved in various gastrointestinal (GI) diseases affecting your digestive system, including:
- Bacterial infections like H. pylori and C. difficile.
- Small intestinal bacterial overgrowth (SIBO).
- Inflammatory bowel diseases, like ulcerative colitis and Crohn’s disease.
- General digestive difficulties, like diarrhea, constipation and gas.
But your gut microbiome also interacts with your brain and with many of your other body systems, including your:
Gut dysbiosis may be indirectly involved in a variety of other conditions, including:
- Malnutrition.
- Malabsorption.
- Food intolerances.
- Irritable bowel syndrome (IBS).
- Atherosclerosis.
- Fatty liver disease.
- Metabolic syndrome.
- Chronic inflammation.
- Chronic fatigue.
- Mood disorders.
Symptoms and Causes
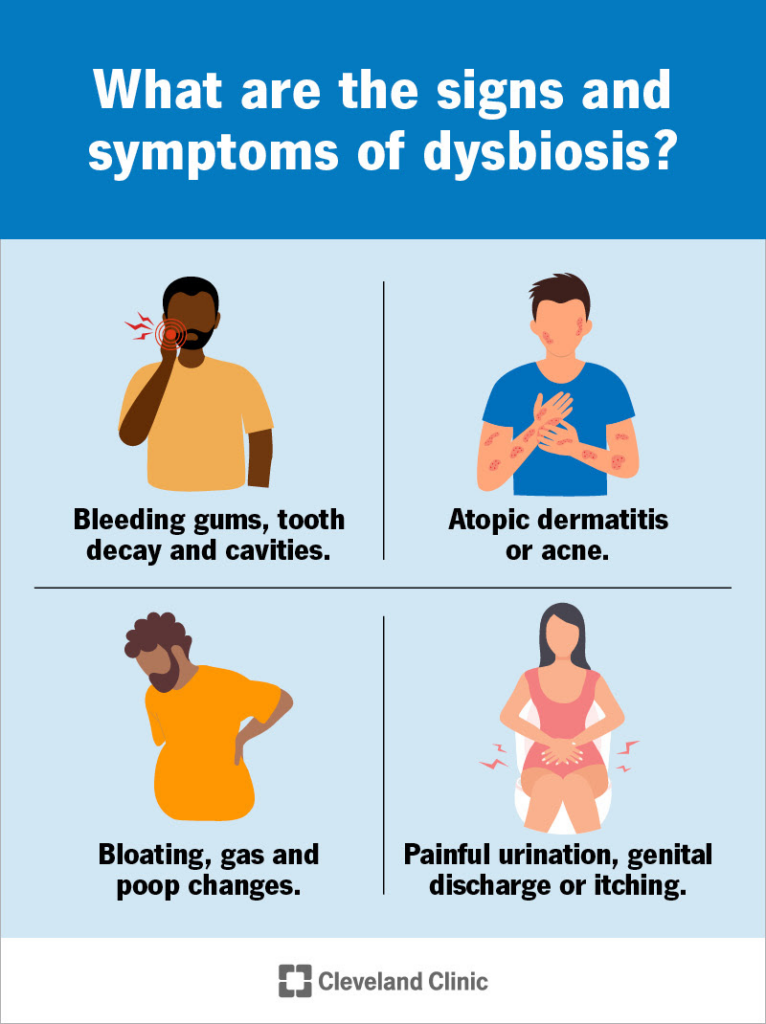
Dysbiosis may cause different symptoms in your gut, mouth, genitals or skin
What are the signs and symptoms of dysbiosis?
You might have dysbiosis in your mouth, on your skin, in your intestine or in your urinary system if you have signs or symptoms of a bacterial, viral or fungal infection or overgrowth there. These might include:
- Bleeding gums, tooth decay and cavities.
- Atopic dermatitis or acne.
- Bloating, gas and poop changes.
- Painful urination, genital discharge or itching.
Intestinal dysbiosis, in particular, may have broader effects — though it’s hard to tell when symptoms outside of your gut relate to your gut microbiome. If you’ve recently developed intestinal symptoms together with other symptoms, like mood changes or weight changes, they might be related.
What causes dysbiosis?
Different things in your internal and external environment can affect your microbiomes, including:
- Antibiotics and antimicrobial agents.
- Other drugs and medications.
- Smoking and alcohol use.
- Environmental toxins.
- Physical and psychological stress.
- Chronic inflammation.
- Chronic diseases.
- Food choices.
Microbiomes are resilient overall, but heavy or long-term exposure to one of these factors, or a combination of several, could cause significant changes. Environmental factors can harm some types of microorganisms while encouraging others, leading to an imbalance. Helpful types of microorganisms might not be able to do their jobs well enough, while the unhelpful types might do more damage.
Diagnosis and Tests
Is there a test to diagnose dysbiosis?
Healthcare providers can diagnose infections with various lab tests. They might take a sample of your blood, poop or urine (pee) or take a swab from your skin, mouth or genitals to test for infection or overgrowth. A pathologist examines the sample in the lab to make the diagnosis. For gut dysbiosis, sometimes a breath test is enough to tell providers about the types of bacteria dominating in your gut.
Management and Treatment
What is the treatment for dysbiosis?
Treatment for dysbiosis depends on the cause. If an underlying disease or condition causes it, you’ll need specific treatment for that condition. If environmental and lifestyle factors contribute to your dysbiosis, your healthcare provider will work with you to change these factors. Environmental and lifestyle changes can usually benefit anyone with dysbiosis, regardless of other causes involved.
If you have an infection or overgrowth, your healthcare provider might need to target it directly with antibiotics, antivirals or antifungals. These medications can diminish the infection or overgrowth, but they can also diminish the helpful types of microorganisms. Some microbiomes will bounce back after treatment, but others might need more follow-up care to help restore their diversity and health.
This might include:
- Probiotics.
- Targeted nutrition.
- In rare cases, fecal microbiota therapy.
Living With
Can I fix gut dysbiosis naturally?
With the right environmental conditions in place, most microbiomes can recover from dysbiosis naturally. You can help it along by making some long-term lifestyle changes, such as:
- Reducing substance use.
- Changing medications if necessary.
- Reducing exposure to environmental toxins.
- Managing stress and addressing its causes.
- Feeding your gut microbiome healthy foods.
- Using probiotics or other supplements, if recommended.
A healthcare provider can help you isolate the factors influencing your microbiome and what it might need to recover. They might recommend specific probiotics or supplements to support your gut health.
What kind of diet helps heal gut dysbiosis?
The simplest way to improve your gut health naturally is to make sure you’re feeding it a diverse and plant-rich diet. Different types of gut bacteria need different types of plant fibers and micronutrients to thrive, so diversity in your diet promotes diversity in your gut microbiome. Whole foods, like plants, also tend to be anti-inflammatory, which makes conditions in your gut friendlier to the friendly bacteria.
Foods to include:
- A variety of whole fruits and vegetables, rich in prebiotic fiber.
- Fermented foods, like yogurt, pickles, miso soup or sauerkraut, rich in probiotics.
- Healthy sources of fats, like fish, nuts and plant oils, which are anti-inflammatory.
Is apple cider vinegar (ACV) with the mother considered better
because it contains more nutrients from the apple, including pectins, malic acid, lactic acid, acetic acid, citric acid, vitamin C, and beta-carotene. “The mother” is probiotic and stimulates the growth of healthy bacteria in the gut. Some studies also suggest that raw, unpasteurized ACV with “the mother” can help lower cholesterol, regulate blood sugar levels, and provide beneficial antioxidants.
Apple Cider Vinegar With Mother Vs Without: What’s The Difference? – Substitute Ninja
Solitude is better for your health when it’s not too intense, research suggests
Can Apple Cider Vinegar Boost Your Gut Health? – GoodRx
Can apple cider vinegar burn away fat cells – Search Videos
Is Apple Cider Vinegar a Probiotic? Here’s the Truth
Does Vinegar Burn and Reduce Body Fat?
Foods to avoid:
- Fast and fried foods, high in inflammatory saturated fats.
- Candy, soda and sweets, high in added sugar.
- Packaged and convenience foods, high in additives and preservatives.
How Sugar throws off Gut microbiome – Search Videos
Articles • By Dr. Jessica Czuba
How Sugar Affects Your Gut and Why It Matters
Your gut is home to trillions of bacteria that help with digestion, immunity, and even mood. But when you eat too much sugar, it can mess with the balance of these bacteria and lead to problems. Here’s how sugar can impact your gut and health:
- Bad Bacteria Thrive on Sugar
Too much sugar feeds harmful bacteria and yeast in your gut, like Candida. This can mess up the balance of good bacteria, causing digestive issues, inflammation, and even contributing to things like anxiety and depression. - Increased Inflammation
Sugar can trigger inflammation in your gut, leading to a condition called “leaky gut.” This means harmful particles can leak into your bloodstream, causing bigger health issues over time. - Nutrient Absorption Problems
Excess sugar can stop your gut from properly absorbing important nutrients like vitamins and minerals. This can leave you feeling drained and lower your immunity. - Impact on Mental Health
Your gut is connected to your brain, so a messed-up microbiome can affect your mood. High sugar diets have been linked to mood swings, anxiety, and even depression.
How to Protect Your Gut
To keep your gut and body healthy, try to:
- Cut back on sugary foods and drinks.
- Eat more fiber-rich foods like fruits, veggies, and whole grains to feed good bacteria.
- Add probiotics (like yogurt or kimchi) to support healthy gut bacteria.
Keeping sugar in check and supporting your gut can help you feel better physically and mentally!
How to eat carbs without spiking your blood sugar, according to a nutrition researcher
Additional Common Questions
Is dysbiosis related to leaky gut syndrome?
Leaky gut syndrome isn’t yet a recognized medical diagnosis. But the theory does involve bad bacteria in your gut. Certain types of gut bacteria will attack and erode your gut lining, weakening your gut barrier. And certain types will produce toxins as byproducts. If these toxins leaked through your weakened gut barrier into your bloodstream, theoretically they might cause an inflammatory response.
A note from Cleveland Clinic
We’re still learning about the many ways that gut dysbiosis might affect our health. Beyond everyday gastrointestinal conditions, your gut microbiome may play a part in a wide range of chronic diseases. We don’t have it all figured out yet. But we do know that diet and lifestyle factors can have a major impact on your gut microbiome. If you think you might have gut dysbiosis, this is a great place to start.
The Role of the Gut Microbiota in Health, Diet, and Disease with a Focus on Obesity
USAID’s foreign aid comes from US farms. Now $450 million of food is left to rot.
Millions of dollars’ worth of American food rots at ports amid USAid shutdown
Dysbiosis in gut microbiome which causes all disease – Search
